%20kopiera%20(1).jpg?width=1920&height=1080&name=f9562214rf%20-%20original%20(924048)%20kopiera%20(1).jpg)
empowering healthcare. improving lives
Efficient digital care
Experience the impact of AI-enabled triage and automation
Right patient, right place, first time
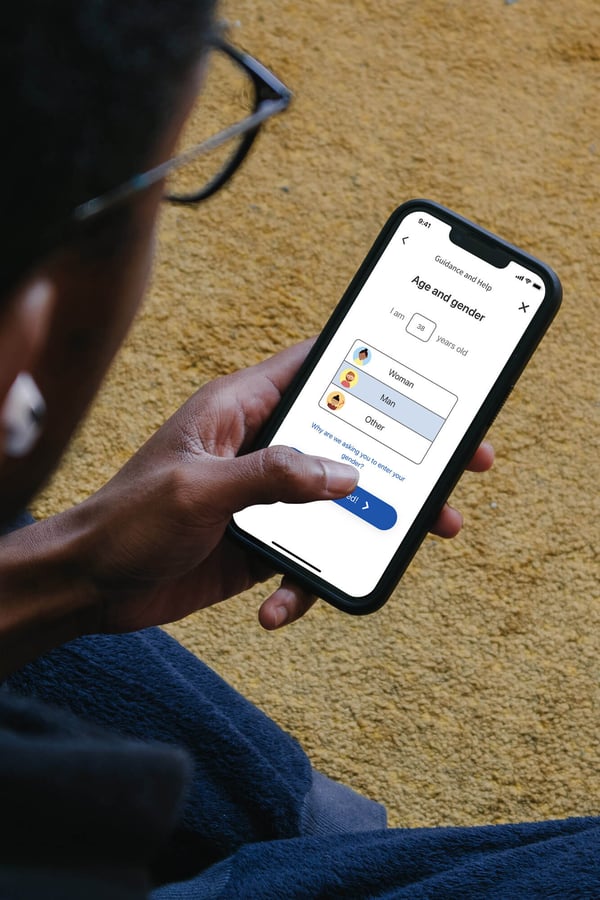
AI-enabled triage tool
Patients can seek help for new symptoms with Red Robin - our AI-enabled triage solution, via your digital front door (website/app) or via an SMS link.
Using free text analysis and intelligent questioning based on probabilistic modelling, Red Robin takes a comprehensive history for new symptoms (including collecting and analysing images) and sends this to the most relevant healthcare professional to manage.
Red Robin also determines:
- a differential diagnosis list with associated probability scores
- a triage/urgency rating based on the Manchester Triage Scale.

Digital front door
Create a single digital front door to your services, empowering patients to seek and manage their own care safely.
Through an app or web-based portal patients can:
- input symptoms/history for new conditions
- book appointments in specific clinics
- request prescriptions
- manage administration requests
- start messaging conversations and more.

Video consultations
Both patients and healthcare professionals see the value of video consultations where appropriate - notably initial screenings and follow-up appointments.
Visiba's video function:
- accommodates up to 10 participants - colleagues, carers, interpreters
- has a waiting room/lobby for you to control who enters and when
- allows screen, chat and file sharing
- alerts the patient regardless of whether they have their patient portal or app open
- has a blurred background option
- includes post call feedback.
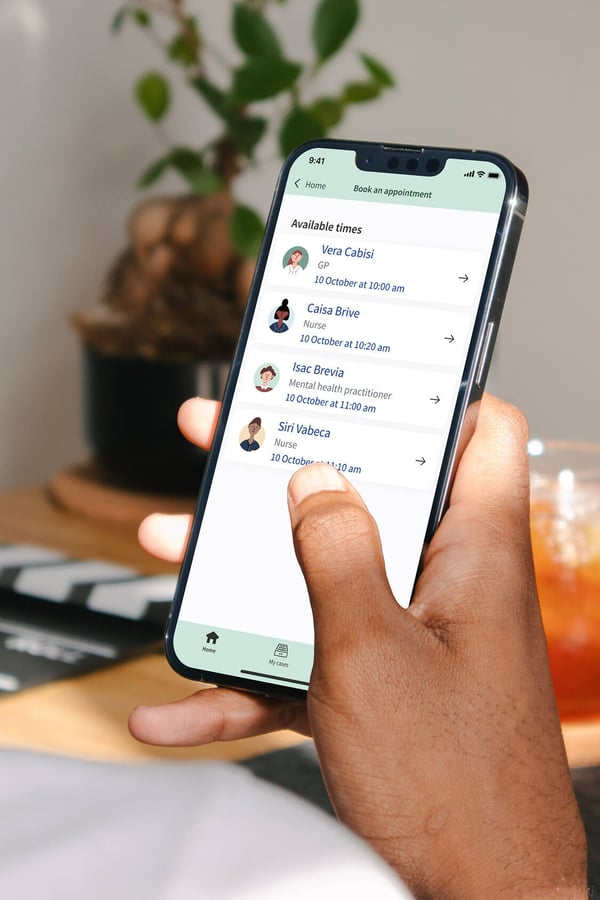
Appointment bookings
Reduce administration by enabling patients to access your services conveniently, in a way that suits them and you. Our platform enables:
- patients to self-book appointments in allocated time slots
- you to invite patients to appointments at specific times
- you to set up digital drop-in clinics
- you to reduce DNAs via reminder notifications (email, SMS or push notifications through the app).
The patient's mobile phone will ring at the time of their appointment - even if they do not have the app open - further reducing DNAs.

Messaging
Communicating securely through messaging is quick and easy with:
- direct secure two-way patient messaging - asynchronously (no set time - like an email conversation) or synchronously (at a set time like a phone call or video chat)
- pre-populated message templates for swift and standardised replies
- attachments such as pictures, videos, forms or files
- secure internal messaging between colleagues
- the option to reassign conversations to other clinics or team members.

Forms and signposting
Forget paper, send patients:
- easy-to-complete digital forms
- digital questionnaires
- links to approved and trusted resources for self-help, information and guidance.
Why Visiba Care?
Continually improving products
Visiba Care is a well-proven solution that has been in use in healthcare since 2014.
Well-tested product enhancements are released every 2-3 weeks, ensuring your Visiba solution will meet your needs now and in the future.
Adoption and implementation
Working closely with our customers we build easy-to-use solutions that improve pathways for patients and healthcare professionals.
Tailored training and support ensures your teams are confident and happy using our solutions.
Clinically validated and trusted
Our medical insights team advise on all product developments to ensure they exceed expectations and are clinically safe and efficient.
Our unique clinical feedback loop means our solution is built on real-world experience.

Security and privacy
Protecting the data of our customers and patients is a top priority in everything we do. We ensure that data sent between servers and clients remain private and encrypted.
We offer 2-factor authentication as standard (including NHS login) with alternative configuration options.
Red Robin is a CE-marked medical device and Visiba Care is ISO 27001 certified and comply with DCB 0129. We consider data protection and privacy issues upfront in all product development to ensure that personal data is processed in compliance with the UK GDPR and the Data Protection Act 2018.
A trusted online consultation platform
2,500
clinics
34,000
HCP users
3million
patient cases

Integrations
We understand that you use multiple systems. Our platform is built for interoperability with them so that you won't have to keep switching between systems or change all of your ways of working to use our solution.
The platform is built on open standards/APIs based on FHIR standards with clear documentation.
Integration options include
- No code - seamlessly connecting flows and functionalities from external systems or content
- Integration modules - where we integrate with other systems via their specific APIs and contracts
- RPA - which can be used to read and write in your system and then interact with our API.

Don't just take our word for it
Why not try it out for yourself?
perspectives
Read our latest articles

Visiba appoints Dr Annabelle Painter as Chief Medical Officer to continue to drive patient safety and clinical efficiency.
Read more
Wakey Wellness – bringing health and wellbeing services to the children and young people of Wakefield
Read more